Over the past year, I have been working on my master’s thesis with the support of The Baird Institute, focusing on understanding the molecular mechanisms underlying thoracic aortic aneurysm (TAA). I’m happy to say that finally my thesis, “Common Proteomic Signatures in Thoracic Aortic Aneurysm: Insights into Underlying Molecular Mechanisms” has been successfully submitted.
TAA is a life-threatening condition and a “silent killer”, characterized by progressive weakening and dilation of the aortic wall, often without warning. Every year, it causes an estimated 150,000-200,000 deaths globally. Current clinical practice relies on measuring the size of the aorta to decide if surgery is needed, yet serious complications often occur below recommended surgery size thresholds. Doctors need better ways to detect the disease, assess its risk, and take action before catastrophic events occur.
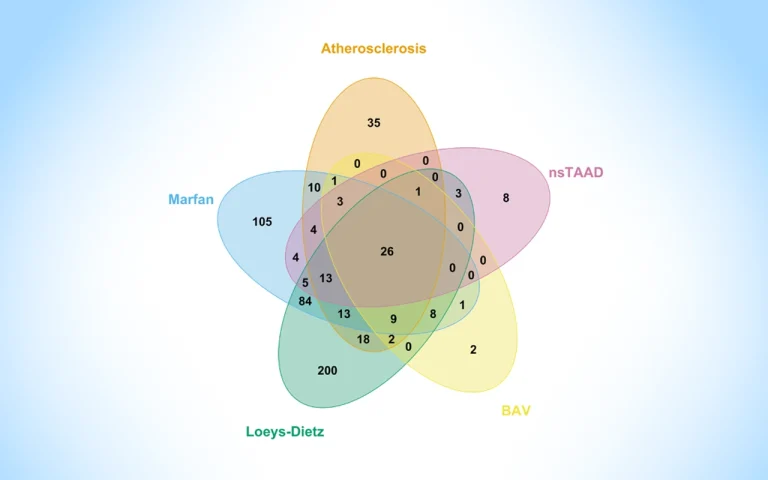
To better capture the underlying biology of disease progression, I studied the proteins in human TAA tissue by comparing healthy aortic tissue with diseased TAA tissue from five different subgroups, degenerative (atherosclerosis), syndromic (Marfan syndrome, Loeys-Dietz syndrome) and non-syndromic (bicuspid aortic valve, familial nsTAAD). The main idea was: “If the same proteins change across all five subtypes of TAA, an underlying problem is shared by all”.
Excitingly, 26 proteins were found altered across all subtypes, either increased or decreased compared to healthy aortas. Many of which are involved in well-known pathological processes already associated with TAAs. Even more interestingly, 14 of these proteins were connected to the TGF-B pathway, one already known to be important in TAA patients with Marfan and Loeys-Dietz syndrome. This discovery could have real clinical benefits. If all TAA subtypes share the same underlying problem, there is the potential for earlier identification of patients-at-risk, better monitoring of disease progression, and hopefully, development of new therapies targeting the shared biological problems found.
This work contributes to the broader effort to transform TAA from a silent killer into a predictable and manageable condition. I look forward to building upon these findings in future research. Next steps will be focusing on strengthening and translating these findings. This research could not have been possible without The Baird Institute and their generous donors, who continue to support and enable future heart and lung research. I’m extremely grateful for the opportunity to contribute to their mission.